|
This results, draw the electrons toward the protons due to greater attraction and decreases the atomic radius. However, there is a greater attraction between the oppositely charged particles protons and electrons. This results in the contraction of the atomic size.Ītomic radius decreases across the periods due to the increasing number of protons. However, the incoming electrons enter the same outermost shell. More number of protons means a more nuclear charge that attracts electrons toward the nucleus of the atom. Protons have positive charges while electrons have negative charges. In the periods, when moving from the left side to the right side, the atomic number increases. Elements are arranged on the basis of their size and nuclear charge. The columns are represented by groups and the rows are periods. Periodic table has seven periods and eighteen groups. It is the radius of the lowest energy electron orbit and can be applied to the atoms or ions that have single electrons like hydrogen, ionized helium, etc. Metallic radius is the distance between the atoms that are bonded by metallic atoms. Ionic radius is the distance or space between the two adjacent oppositely charged ions and the length of the ionic bond should be equivalent to the sum of their ionic radii. This is the radius between the two covalent bonded atoms and the length of the covalent bond must be equal to the sum of their covalent radii. This is because metals have van der Waal’s interactions that are generated by the quantum fluctuations of atomic polarisation. In metals, van der Waal’s radius is mostly defined for metals. It is a type of atomic radius defined as the minimum distance between the nuclei of two atoms that are not bounded by covalent or metallic bonds. These are the following widely used definitions of atomic radius: It decreases across the periods from left to right while increasing down the groups from top to bottom. However, atomic radius trends vary with periods and groups in the periodic table. It has different values for different atoms depending on their sizes. These trends explain the periodicity observed in the elemental properties of atomic radius, ionization energy, electron affinity, and electronegativity.Atomic radius is the distance from the nucleus of an atom to its outermost shell or electron. This happens because the number of filled principal energy levels (which shield the outermost electrons from attraction to the nucleus) increases downward within each group. Second, moving down a column in the periodic table, the outermost electrons become less tightly bound to the nucleus. As this happens, the electrons of the outermost shell experience increasingly strong nuclear attraction, so the electrons become closer to the nucleus and more tightly bound to it. First, electrons are added one at a time moving from left to right across a period. 
In addition to this activity, there are two other important trends. Stable octets are seen in the inert gases, or noble gases, of Group VIII of the periodic table. Elements tend to gain or lose valence electrons to achieve stable octet formation. These trends can be predicted merely by examing the periodic table and can be explained and understood by analyzing the electron configurations of the elements. 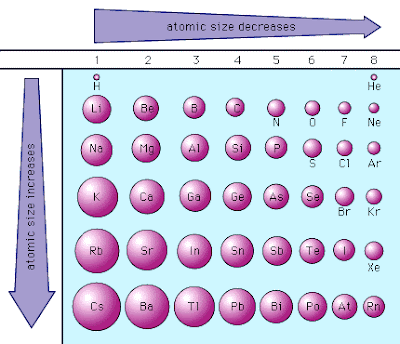
The periodic table arranges the elements by periodic properties, which are recurring trends in physical and chemical characteristics.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
